In this age of technology, there is a development every minute in almost all the fields in one regard or the other. Such a significant result in a lot of alternate energy is the Fuel cell. A fuel cell, in simple terms, is an electrochemical energy generating device. It has been one of the most fascinating and interesting aspects of today’s world’s technology. Many things are yet to be developed in this field, and there are a huge scope and application for these Fuel Cells. These fuel cells are being under research by many people all around the world.
Construction and Working
There are many different types of fuel cells. But the basic construction and working of a fuel cell are the same, and the concept behind it is also the same. The Fuel cell works on the principle that energy is produced when some chemical reaction occurs. If this energy can be tapped, it can be converted into practical means. In a conventional fuel cell, the reaction occurs between hydrogen and oxygen, and water is formed. This is the primary chemical reaction that happens, and energy produced by this is tapped through proper channels and used for several applications. It is a unique chemical reaction that is being the interest of many people. The by-product is water, and hence fuel cells are environmentally very safe to use.
The conventional fuel cell consists of a fuel chamber, an oxidant chamber, and the reaction chamber. The fuel and oxidant react in the reaction chamber, which has an electrolyte and produces energy in a slight current. This current is tapped and used. Similar stakes of these sets are connected in series to make an appreciable quantity of wind.
Types and Applications
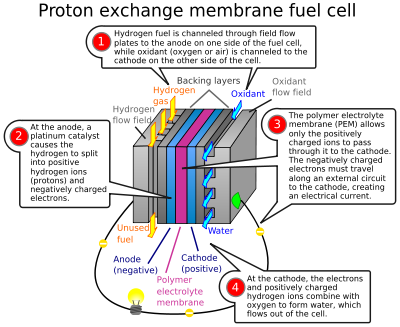
Depending on the working conditions, electrolyte, the quantity of energy produced, fuel cells can be divided into many types. They include types like metal hydride fuel cells, proton exchange fuel cells, alkaline fuel cells, etc. Of these, the Proton exchange membrane (PEM) fuel cells are of primary concern to many. It has the correct working temperature range, easy availability, simple construction of all others. Here a polymer membrane acts as the electrolyte, and there is a Platinum catalyst to speed up the reaction. The primary application of these fuel cells in the automotive industry. They are also used in several other fields, but this automobile industry is their direct application.
Scope and Current Position
There is a lot of scope for these fuel cells in many applications. The current position of the fuel cell is that they are still in the research stage. Many developments have occurred over time, but there are still many scopes for development. This can be well explained by citing the fact that there is not enough platinum left in the world that could meet all people’s demands. So there is research going on to see the availability of other materials.
Advantages and Disadvantages
There are many advantages like its environment-friendly working, easier installation, simple working mechanism, and low maintenance. But it has some disadvantages as well. They are its cost, less life, rare availability of some elements in the cell, and other technical difficulties that have made these excellent prospects still in the development stage. It can be believed that someday, they can be commercialized in a huge quantity, and the common man can have access to them.


I was eating poptarts when I came across this here article. I would like to know, Are Poptarts Made Out Of Recycled Fuelcells? And, are they safe for my 2 year old son to eat??
can this here feul cell thingy majigy help me take over the world. If not can I make a sandwich with it.
if i drink the fluid from a fuell cell will it quench my thirst. if not can i use it for toilet water.
i was on my porch eaten fried chicken and drinkin coolaid when i came across this article me and my clan members saw it as an oportunity for our new wapower float will ity power it?
Who ever this \Mr. Jone$$\ person is, you are truley an idiot. Your computer is being tracked right now. Soon the police will be there. Congrats smarty.
I was singing about my fake girlfriend when I read your article it looks like a toaster. I would like to make your website better by singin my song on your here web.
queers
http://www.fuelcellenergy.com/benefits-fuel-cell-technology.php